Or maybe I will move on to drinking smoothies that won’t spill as much as coffee. I think adding honey will help thicken it up a bit. Wow!Īfter learning about the viscosity of liquids, I know why my coffee spilled everywhere! Since, it contains water, it follows the properties of water. As a scientist I might say: an increase in the viscosity of blood can severely decrease the blood flow to the fingers, thus my fingers are cold. If blood moves slower it doesn’t move through the body as quickly the blood is less able to do its job keeping us warm. But have you ever forgotten to wear gloves or a coat in cold weather? If your fingers or body grew cold, that means the temperature of your blood became cooler too. The body’s core temperature is maintained by the blood moving throughout. But what if our body temperature drops? This will make the blood more viscous, which means it will flow more slowly. 
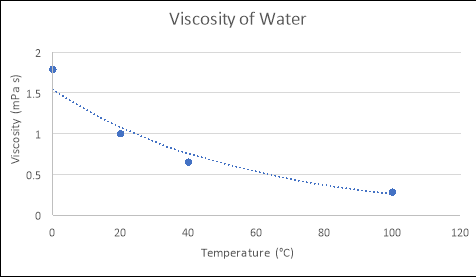
Our blood moves and keeps us warm or helps us cool down. It is responsible for transporting things like nutrients, oxygen and immune cells. Think about blood, an integral part of our body. Liquids are not just things we drink or find in the world. What happens when honey is poured into a syringe? What if a plunger is placed within the syringe? The pressure in the syringe increases and in turn increases the viscosity of the honey. Whereas, an increase in pressure, will lead to an increase in a liquid’s viscosity. I bet you found that heated honey flows more easily than colder honey. What if you heat the honey? Think back to when you were baking and used honey. An increase in the temperature will lower the fluid’s viscosity. Temperature also plays a role in changing fluid viscosity. Also the bigger a molecule is in size, the more viscous it becomes. The stronger the bonds are within a molecule, the harder it is for the molecule to move and therefore the more viscous it is. One factor is the bonds that bind a molecule within a liquid. Science behind viscosityĪ variety of factors can affect viscosity. Whereas, a substance like water, when poured, flows faster than honey, and is therefore less viscous. This means it has high resistance to flow and is therefore highly viscous. A substance like honey, when poured, flows slowly. What if you poured each one into a container? How do you think they will flow? Fast? Medium? Slow? This variation is viscosity. Viscosity of a fluid is “a measurement of its resistance to flow.” It is measured in poise (P) or centipoise (1cP = 10 -3 P). Liquid has many properties – vaporization, condensation, boiling point, critical temperature and pressure, surface tension and viscosity. What different liquids have you used, consumed or come across during the past week? What was different about them? Did they have different colors? Were they fizzy or still? Did they feel thick or thin? What about pouring? How did each one flow? So, much time would be saved in cleaning! This made me wonder about properties of liquids that make them flow. It went everywhere! While rushing around trying to find paper towels, I wondered – wouldn’t it be great if the coffee didn’t spill everywhere. Yesterday, while working on a paper, I spilled a cup of coffee.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
